|

DEPARTMENT OF PHYSICS AND
ASTRONOMY
LINKS
|

Basics: Metallic Quantum Wells
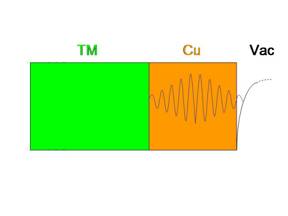 When
the dimension of a metallic system enters the nanoscale regime, its
electronic structure acquires a discrete component to its spectrum.
These quantum size effects are ubiquitous and mediate phenomena such
as the interlayer exchange coupling in magnetic multilayers, magic
numbers in thin film and island growth, conductance anomalies in
epitaxial films and chemisorption properties. The formation of
so-called metallic quantum well (MQW) states is schematically
illustrated at the left. Significant electron reflectivity at the
Cu/transition metal (TM) interface causes states to be confined to
the Cu overlayer. These states change energy as the thickness of the
Cu overlayer increases and modify the density of states at the Fermi
level as they pass through it. When
the dimension of a metallic system enters the nanoscale regime, its
electronic structure acquires a discrete component to its spectrum.
These quantum size effects are ubiquitous and mediate phenomena such
as the interlayer exchange coupling in magnetic multilayers, magic
numbers in thin film and island growth, conductance anomalies in
epitaxial films and chemisorption properties. The formation of
so-called metallic quantum well (MQW) states is schematically
illustrated at the left. Significant electron reflectivity at the
Cu/transition metal (TM) interface causes states to be confined to
the Cu overlayer. These states change energy as the thickness of the
Cu overlayer increases and modify the density of states at the Fermi
level as they pass through it.
|
Recent Highlights: Metallic Quantum Wells
Chemisorption modified
by quantum confinement of electrons
As suggested by the discussion above, as the thickness of a metallic
quantum well changes, MQW states change energy. The figure below on
the left shows a series of inverse photoemission spectra obtained
from the Cu/fccFe/Cu(100) system as a function of increasing film
thickness. The tick marks show that MQW states move upward with
increasing thickness. In particular, there is a strong
modulation of the intensity, reflecting a modulation of the density
of states (DOS) as MQW states pass through the Fermi level (EF).
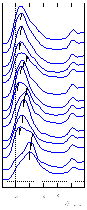
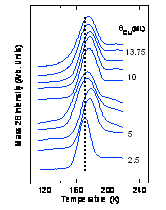 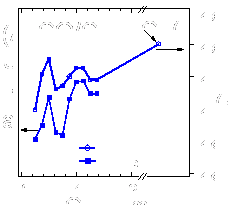
The center figure above shows
carbon monoxide (CO) temperature programmed desorption spectra from
Cu MQWs grown on top of the fccFe films. The peak desorption
temperature (TD) is a measure of the strength of the
CO-metal bond. The non-monotonic change in TD shows
that the CO-MQW bond strength modulates as a function of MQW
thickness. The figure above on the right shows that there is a
direct correlation between changes in TD and
modulations in the DOS at the EF caused by the MQW
states. These results demonstrate that quantum confinement of
electrons is a viable way to modify chemisorption at surfaces, and
may be used to modify surface reactions, catalysis, electronic
friction, or molecular self assembly.
Read more:
Quantum size effect induced modification of the chemisorption properties of thin metal films
; A. G. Danese, F. G. Curti *, and R. A. Bartynski, Physical Review B
70, 165420 (2004)
|
Recent Publications
(back to top)
-
Carbon monoxide adsorption on the fccCo/Cu(100) metallic quantum well system
L. Tskipuri and R.A. Bartynski, Surface Science 603, 802 (2009)
-
Room temperature ferromagnetism in Mn ion
implanted epitaxial ZnO films
D.H.Hill, D.A. Arena,
R.A. Bartynski, P. Wu, G. Saraf, Y. Lu, Wielunski, R. Gateau, J.
Dvorak, A. Moodenbaugh, and Y.K. Yeo, Physica Status Solidi A,
203, 3836 (2006)
-
Ferromagnetism in Fe-implanted a-plane ZnO
Films
D.P. Wu, G. Saraf, Y. Lu, D.H. Hill, D.A. Arena, R.A. Bartynski, L.
Wielunski, R. Gateau, J. Dvorak, A. Moodenbaugh, T. Siegrist, J.
A. Raley, and Yung Kee Yeo, Appl. Phys. Lett.89, 12508
(2006)
-
The relation between crystalline phase,
electronic structure, and dielectric properties in high-k gate
stacks
S.
Sayan, M. Croft, N.C. Nguyen, T. Emge, J. Ehrstein, I. Levin, J.
Suehle, R.A. Bartynski, and E. Garfunkel, AIP Conf. Proc. 788,
92 (2005)
-
Dichroic effects in Auger-photoelectron
coincidence spectroscopy of solids
R.
Gotter, F. Da Pieve. A. Ruocco, F. Offi, G. Stefani, R.A.
Bartynski, Phys. Rev. B. 72, 235409 (2005)
-
Inverse Photoemission Spectroscopy from
Al(100)
J. F. Veyan, W. Ibanez, R.A.
Bartynski, P Vargas, and P. Haberle, Phys. Rev. B. 71,
155416 (2005)
|
|